How To Find The Most Reactive Position In Electrophilic Aromatic Substitution Reactions?
- Jan 25, 2022
- 1 min read
Updated: Nov 21, 2022
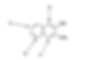
Predict which position of the naphthalene compound below is the most reactive with electrophiles in electrophilic aromatic substitution?