A hydrogen atom electron in an excited state with n = 7 drops to a lower energy state with n = 3. What is the wavelength of the light emitted in this transition?
- Feb 14
- 1 min read
A hydrogen atom electron in an excited state with n = 7 drops to a lower energy state with n = 3. What is the wavelength of the light emitted in this transition?
Solution:
To calculate the wavelength of light emitted when an electron travels from n=7 to n=3, we have to use Rydberg's equation.

Where R is the Rydberg's constant, nf is the final energy level, ni is the initial energy level, h is Planck's constant, c is speed of light and λ is wavelength.
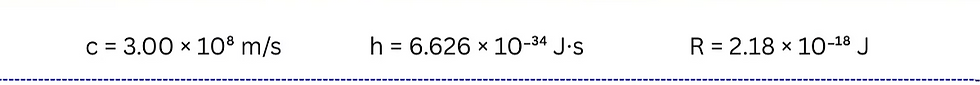
We will have to take absolute value of the energy we find so solve for lambda. −2.18 × 10⁻¹⁸ J (1/3² − 1/7²) = 6.626 × 10⁻³⁴ J·s)(3.00 × 10⁸ m/s)/λ
λ= 1.005 × 10⁻¹¹m ( 10⁹nm/1m) = 1006 nm.
LINKS:

Comments